Clean-in-place (CIP) processes are essential to industries such as food and beverage, personal care products, and any other batch processes where residual contamination can negatively impact product quality.
Critical factors in clean-in-place management
Time
Time spent conducting CIP cycles is time that is not spent on production, so CIP cycles should be as short as possible while still being effective.
Chemicals
Under-dosing of cleaning chemicals increases the risk of contamination and biofilm formation and will also necessitate additional chemical treatments to mitigate these more serious issues later. Conversely, over-dosing of chemicals also carries a significant cost and can upset downstream wastewater treatment processes.
Water
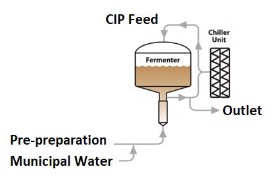
Depending on the size of vessels and lines that require cleaning, water requirements can be excessive. These costs can be compounded by the amount of energy and chemical needed when water usage increases.
Energy
The costs associated with pumping as well as heating of water and cleaning agents is directly tied to the amount of water and chemical required. Considering that electricity costs are often among the largest expenses in operating industrial processes, these costs can escalate quickly if water and chemical usage is not optimized.
How can the effectiveness of a CIP cycle be quickly verified?
A holistic water quality monitoring toolbox including rapid microbiological monitoring enables all aspects of CIP cycles as well as the production process as a whole to be optimized.
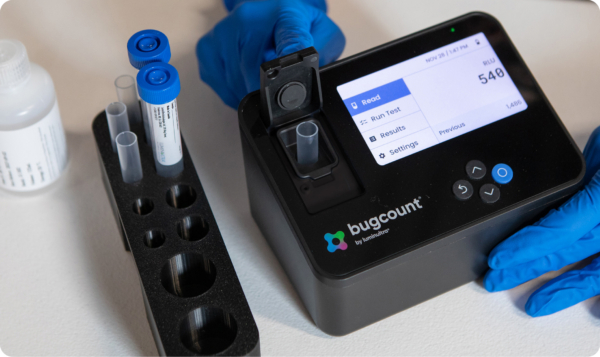
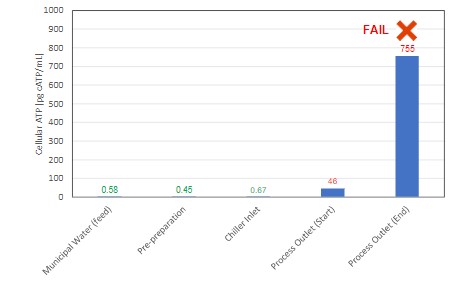
ATP monitoring has been used for decades in food processing to quickly assess surface cleanliness. While this technology still serves an important role, 2nd Generation ATP focuses on analysis of water and other aqueous samples. In doing so, it greatly expands the applications for this rapid method and enhances QA/QC programs to a significant degree.
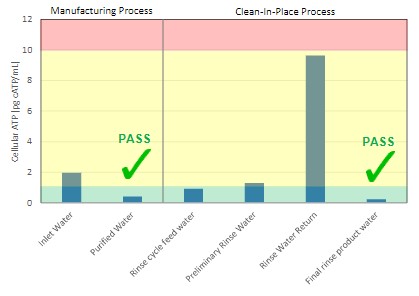
Example 1: Food processing QA/QC