Safeguard your system uptime
Commercial and industrial operations depend on large volumes of water to function, whether it’s for cooling, production or treatment. But without visibility into what’s happening in that water, it’s hard to take action when it matters. Luminultra helps you monitor microbial activity in real time, keeping your systems running efficiently, protecting your equipment and safeguarding the health of your communities.
01 Legionella management
Problem
Preventable outbreaks still threaten public health
Legionella is one of the most significant sources of waterborne disease outbreaks in the United States. It’s also largely preventable: investigations by the CDC indicate that many cases could have been prevented through better water management.
Solution
Fast detection with end-to-end support
Luminultra’s suite of microbial monitoring tools equip you with everything you need to quickly and accurately detect Legionella in cooling towers. With scalable solutions that work from small scale to the largest operations, including datacenters, you can maintain confidence in the health and safety of your communities.
Where this matters
- Data centers
- Industrial manufacturing
- Health care and long-term care facilities
- Commercial properties
- Hospitality
Explore more
02 Microbial contamination
Problem
Microbes can derail critical processes
In industrial operations, water is essential for heating, cooling, rinsing and chemical processing. When microbes contaminate systems like make-up water or slurries, they can disrupt production, degrade product quality and trigger costly downtime.
Solution
Real-time insights for faster intervention
Luminultra’s microbial monitoring tools help you detect, segregate and remediate biological growth within a product before issues materialize. Identify biological growth early and maintain confidence in your control programs with fast, reliable testing.
Where this matters
- Make-up water and open recirculation systems
- Slurry and chemical feed system oversight
- Paper machine and pulp processing
- Wastewater and effluent monitoring
- High-purity and ultrapure water systems in semiconductor facilities
- Rinse tanks and paint line microbial control in automotive plants
Explore more
03 Efficiency loss and biofilm buildup
Problem
Microbial build-up is costing the bottom-line
Microbial build-up causes biofilms which are harming the efficiency of your processes. Even if not causing an upset, undiagnosed and untreated microbial activity means your system isn’t operating as it should. In the scale and size of today’s industrial operations, these are costly issues that continue to worsen over time.
Solution
Detect, control and prevent
Luminultra’s fast, accurate microbiological testing and monitoring tools help you stay ahead of biofilm formation and microbial-induced inefficiencies. By identifying microbial activity in real time, you can pinpoint problem areas, optimize treatment programs, and prevent long-term damage to your systems.
Where this matters
-
Cooling towers and HVAC systems
-
Boiler and steam systems
-
Process water loops
-
Storage and distribution networks
Explore more
04 Biocide management and optimization
Problem
Chemical treatment shouldn’t be guesswork
Biocide programs are essential for controlling microbial growth in industrial water systems. But without reliable monitoring, treatment often relies on trial and error—leading to overdosing, underdosing, or missed problems that escalate into costly failures.
Solution
A smarter way to control biocide use
Luminultra’s microbial monitoring solutions give you real-time insight into system health, so you can apply the right amount of biocide at the right time. In just minutes, you get accurate, actionable results that complement your existing control programs and reduce waste, risk, and downtime.
Where this matters
- Cooling and heating system treatment
- Closed-loop and evaporative cooling systems in datacenters
- Process water microbial control
- Slurry and rinse water management
- Chemical dosing program validation
- Water-based equipment maintenance
- Continuous monitoring for regulated environments
Explore more
Not sure what you need? Let’s figure it out together
Talk to a Luminultra expert and get clear answers, fast.

Build your complete microbial toolkit
Test smarter. Act faster.
Access a full range of solutions designed to bring confidence to the way you work.
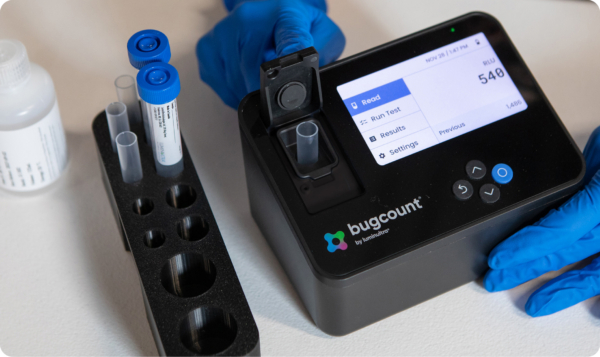
From rapid field testing to advanced lab analysis, Luminultra delivers the insight you need to stay ahead of microbial risk. Whether you're using Bugcount® 2nd Generation ATP, Genecount® qPCR, our extensive lab services or our digital platform, Relay™, our solutions put you in control.
What our clients are saying
Real stories of challenges solved with confidence.